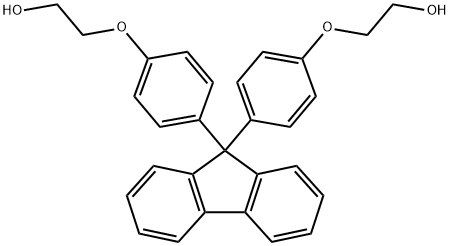
Bisphenoxyethanolfluorene: Optical Applications & Antiestrogenic Activity
Bisphenoxyethanolfluorene (also name 4,4′- (9-Fluorenylidene)bis (2-phenoxyethanol); BPEF) is a low-mass compound, primarily used in the preparation of polymer resins and films.Its core application lies as a monomer for synthesising high-performance polymers such as epoxy resins, polycarbonates, polyesters. Downstream products find extensive application in optical resins, functional film materials, electronic encapsulation materials, and aerospace materials.

Incorporation of low-mass compound to alter the orientation birefringence
Cellulose esters are biomass-derived materials that have found their applications in various fields. In an optical film application, some cellulose esters such as cellulose triacetate (CTA) and cellulose diacetate (CDA) have been utilized as a photographic film base and a polarizer protection film for years. The excellent inherent properties such as high transparency throughout the visible wavelength range and high heat resistance make cellulose esters suitable for optical film application. Furthermore, it has been found that the optical and mechanical properties of cellulose esters can be modified by blending with other polymers or plasticizers. The orientation birefringence of CAP added with bisphenoxyethanolfluorene (BPEF) is discussed in this study. Our results show that the orientation birefringence of hot-drawn CAP films can be reduced by Bisphenoxyethanolfluorene addition. Furthermore, the results are compared to that obtained from the CAP/TCP blend. All sample films were prepared using melt mixing method. The orientation birefringence of CAP containing the LMC is discussed by considering the effects of (i) degree of orientation and (ii) structural unit of polarizability anisotropy. Furthermore, the effects of drawing temperatures and draw ratio on the orientation birefringence of the LMC are studied to evaluate the nematic interaction.[1]
Both Bisphenoxyethanolfluorene and TCP are found to be miscible with CAP as indicated by a single peak ascribed to the glass-to-rubber transition in their respective E′′ curves. In particular, the peak height of the CAP/BPFE blend is almost identical to that of the pure CAP, suggesting a good miscibility of the system. An abrupt drop of the storage modulus E′ owing to the glass-to-rubber transition is observed for all samples (not shown here).Understanding the mechanism by which a low-mass compound (LMC) influences the orientation birefringence in cellulose esters is important in designing and producing an ideal film that fulfills its optical requirements. In this study, the effect of the LMC addition on the orientation birefringence as well as on its wavelength dispersion is evaluated. The orientation birefringence of CAP blended with different types of LMC, i.e., Bisphenoxyethanolfluorene and TCP, is measured and then analyzed in comparison to that of pure CAP. Addition of the LMC affects the magnitude as well as the wavelength dependence of orientation birefringence. Bisphenoxyethanolfluorene addition decreases the orientation birefringence, indicating the increase in polarizability anisotropy perpendiculars to the stretching direction. Furthermore, it is effective in enhancing the extraordinary wavelength dispersion behavior of the orientation birefringence. Lastly, it is found that Bisphenoxyethanolfluorene shows a larger orientation birefringence than TCP, despite the values of their intrinsic birefringence are opposite. This indicates that the nematic interaction between BPEF molecules and CAP polymers is higher that that of TCP.
Antiestrogenic property of Bisphenoxyethanolfluorene
In recent decades, many endocrine-related diseases and disorders have been increasing (WHO, 2013). Identifying the related endocrine disrupting chemicals (EDCs) and managing their uses are necessary Because the chemicals recognized as EDCs are only the “tip of the iceberg” and because many chemicals in consumer products are not assessed by the manufacturer, identifying chemicals with endocrine disrupting properties linked to disease outcomes is a real need. Bisphenoxyethanolfluorene has been widely and increasingly used as a monomer or an additive to synthesize polymers in optical products since 1990 s. To date, highly functionalized polymers derived from BPEF, such as dimethacrylate resins
, polyester, polyurethane
, cellulose ester
, epoxy acrylate
, and polycarbonate resin (
Kato et al., 2017), process superior optical, thermal and mechanical properties. Currently, materials containing Bisphenoxyethanolfluorenes are widely used in elastomers, coatings, sealants, adhesives photographic film base, polarizer protection film, liquid crystal display industry, digital camera lenses, and dental composite materials. Inevitably, human body would expose to Bisphenoxyethanolfluorene during its release from polymers in the production and usage processes of these commodities, as is known that bisphenol A and other bisphenols, phthalates, etc., can be released from plastic drink bottles, toys and other products containing these chemicals.[2]
BPEF has been widely used to produce fluorene-containing polymers for several decades, and pollution of this chemical to the environment and humans during production process or product use is inevitable. We believe that our data suggest a compelling need for investigation of Bisphenoxyethanolfluorene levels in the environment and humans. Here, we reported the antiestrogenic activity of BPEF by in vitro, in vivo and in silico approaches. The antiestrogenic activity of BPEF in vitro is so strong which is similar to that of tamoxifen, the antiestrogenic drug. BPEF inhibited the uterine development and caused endometrial atrophies at relatively low doses in adolescent mice after 10-d treatment, and downregulations of BPEF on these estrogen-responsive genes in the uteri of mice might be the underlying mechanisms. BPEF could fit well into the antagonist pocket of human ERα, and the formation of hydrogen bonds and hydrophobic interactions between chemical and the active site of receptor maintained their strong binding. Considering that Bisphenoxyethanolfluorene possesses strong antiestrogenic property and can disrupt female development, we suggest that it should not be used in products that are in close contact with people, particularly immature women. Because no exposure levels have been reported for BPEF so far, we believe that our data indicate a compelling need for investigation of BPEF levels in the environment and humans.
References
[1] Mohd Edeerozey Abd Manaf . (2013). Incorporation of low-mass compound to alter the orientation birefringence in cellulose acetate propionate. Optical Materials, 35 7, Pages 1443-1448.
[2] Jia, Xiaojing et al. “Antiestrogenic property of 9,9-bis[4-(2-hydroxyethoxy)phenyl]fluorene (BPEF) and its effects on female development in CD-1 mice.” Ecotoxicology and environmental safety vol. 242 (2022): 113906. doi:10.1016/j.ecoenv.2022.113906
You may like
Related articles And Qustion
See also
Lastest Price from Bisphenoxyethanolfluorene manufacturers
US $0.00/kg2026-03-20
- CAS:
- 117344-32-8
- Min. Order:
- 1kg
- Purity:
- 99.5%
- Supply Ability:
- 20tons
US $0.00/kg2025-12-10
- CAS:
- 117344-32-8
- Min. Order:
- 25kg
- Purity:
- ≥99.5%
- Supply Ability:
- ton